A new antibody cocktail developed by Israeli researchers may provide natural immunity against COVID-19 for weeks, and possibly months, according to a preliminary study.
The recently released study, published last week in bioRxiv and not yet peer-reviewed, was conducted by a team of Israeli scientists led by Dr. Natalia Freund and PhD student Michael Mor at Tel Aviv University’s Sackler Faculty of Medicine.
The team isolated and characterized six antibodies derived from the blood of severely ill COVID-19 patients, and then combined three antibodies at a time for a potent in-vitro cocktail against SARS-CoV-2, the virus that causes COVID-19, according to a university announcement. The scientists have filed for a patent of the antibodies through Ramot, the Technology Transfer Arm of Tel Aviv University. Their study is also currently under consideration by the peer-reviewed medical journal PLOS Pathogens.
SEE ALSO: Israel Set To Launch Human Trials For COVID-19 Vaccine After High Holidays
“The antibodies bind to the virus in different spots and neutralize the virus using different mechanisms,” Dr. Freund explains to NoCamels in a phone interview. The antibodies “were more effective when combined as opposed to by themselves, and we got very efficient neutralization of the virus in tissue culture.”

Pending successful clinical trials with humans, the data from the study support “the use of combination antibody therapy to prevent and treat COVID-19,” the scientists wrote in their study.
The quality of antibodies
The study began in April, approximately a month into Israel’s first national coronavirus lockdown. The team sought to understand the antibody response to COVID-19 infection and the development in patients with no symptoms, mild symptoms and severe symptoms “with regard to both the quality and quantity of the anti-viral antibodies produced by the immune system.”
The scientists recruited 18 of Israel’s earliest COVID-19 patients – 10 with mild or no symptoms, and eight severely ill patients who were hospitalized, and some even ventilated, at Ichilov and Kaplan Medical Centers. (All the participants recovered from the disease.) They found that only a small portion of the mildly ill participants developed neutralizing antibodies, and some developed none. Meanwhile, the blood of all severely ill patients contained neutralizing antibodies.
These findings suggest that those with a mild or asymptomatic infection may possibly contract the disease a second time, while those who recovered from a more severe infection may be protected, according to the university statement.
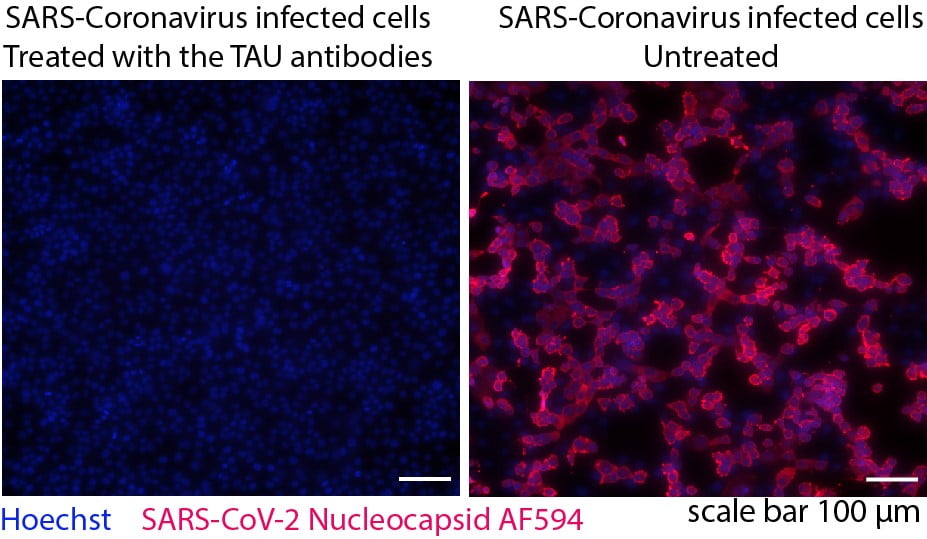
“We sequenced the cells that are producing these antibodies; these cells are called B lymphocytes, these are cells of the immune system,” Dr Freund explains, emphasizing that they are found naturally in the body.
“We sequenced thousands of these cells, and eventually we were able to isolate six different antibodies that were neutralizing [of the virus] and produced them in the lab. So we basically reproduced the antibodies and made a monoclonal (lab-made identical immune cells that are all clones of a unique parent cell) antibodies, and we mixed these and tested their use in neutralizing the virus, alone and in combination with each other,” she tells NoCamels.
Antibody cocktail
The concept, she says is the same as the experimental antibody cocktail administered to US President Donald Trump earlier this month when he was found to have tested positive for COVID-19 and began developing symptoms.
The cocktail treatment the president received was developed by American biotech company Regeneron; early studies have shown that it can be effective in patients whose immune systems had not mounted their own antibody response. Trump himself called the treatment a “miracle” and has touted it as a cure, but the cocktail has not yet been fully clinically evaluated.
Sign up for our free weekly newsletter
SubscribeThe Israeli scientists’ cocktail, explains Dr. Freund, is derived naturally by the patients’ immune systems, which means that it is probably safer for use “because it’s natural human antibodies, and there are no expected adverse effects,” she tells NoCamels. “Since these antibodies are stable in the blood, a preventive injection can provide protection for several weeks, and possibly even several months.”
SEE ALSO: Early Results For COVID-19 Trial With Antibody Drug Show 90% Improvement Rate
This type of treatment also differs from other antibody therapies such as those derived from plasma.
“We are not taking the antibodies from the plasma and purifying them and using them on patients, or it’s not our intention to,” says Dr. Freund.
The vision is that, in the future, the cocktail will be used to treat COVID-19 patients, “until the much-awaited vaccine finally arrives,” says Dr. Freund.
COVID-19 vaccine development
Currently, close to 200 candidate vaccines or treatments for COVID-19 are in development, 42 of which are in clinical evaluation as of October 2020. These include a promising vaccine candidate developed by Massachusetts-based company Moderna, which is currently in Phase III trials, and a vaccine developed by the University of Oxford which signed a distribution agreement with drugmaker AstraZeneca
In August, Israeli researchers from the government-run Israel Institute for Biological Research (IIBR) indicated that they expect to begin human trials for the COVID-19 vaccine candidate they developed after the high holidays.
The institute announced several developments over the past several months. In June, researchers indicated that a vaccine they developed for SARS CoV-2, the virus that causes COVID-19, was found to be effective in trials involving hamsters, paving the way for testing with humans. Previously, the center reported “significant progress” on the vaccine and initial trials.
The secretive institute has also been working on researching potential treatments and in early May announced that it made a breakthrough on a treatment involving a discovered antibody that neutralizes the virus. That same month, it further announced that researchers found that a combination of two existing antiviral drugs for Gaucher disease appears to inhibit the growth of SARS CoV-2, and may work against other viral infections, including a common flu strain.
On Sunday, the Hebrew-language Walla news site reported that clinical trials for the IIBR vaccine may begin later this month, pending the approval of the Health Ministry and that of the Helsinki Committee, a medical panel comprised of physicians and advocates that weighs research approval for human experiments.
The study is expected to unfold in three stages: the first will consist of a trial involving a hundred healthy participants aged 18-50; the second will consist of 1,000 participants and is expected to start in December; the third stage will involve some 30,000 volunteers and may begin early next year.
Related posts

Israeli Medical Technologies That Could Change The World

Harnessing Our Own Bodies For Side Effect-Free Weight Loss

Missing Protein Could Unlock Treatment For Aggressive Lung Cancer




Facebook comments