This article was written for NoCamels by ZAVIT – Science and Environment in Israel
There’s an old saying that the way to a person’s heart is through their stomach, and today, the way to a healthier quality of life is — quite literally — by way of the gut.
Israel is known for being one of the world’s healthiest countries, due in part to the Mediterranean diet, rich in vegetables, fruits, nuts, legumes, and healthy fats like olive oil. The average Israeli has access to approximately 152 kg of fruit and 154 kg of vegetables each year, according to the Israeli Central Bureau of Statistics, and 80 percent of Israelis eat fresh fruit and/or vegetables daily.
As is customary in Israel, incorporating fresh salads into large breakfasts works to regulate blood sugar, facilitate weight loss, and avoid insulin resistance for diabetics. In other cases, this diet has been shown to prevent chronic and degenerative diseases. Even when the diet was tested on patients who had previously suffered from heart attacks, the occurrence of acute coronary episodes was reduced by 50 percent.
SEE ALSO: Israel In Numbers: Snapshot Of A (Largely) Happy, Healthy, Educated Nation
The benefits of a Mediterranean diet have piqued scientists’ interest in individual gut health and personalized medicine and nutrition, all based on a patient’s individual characteristics. This growing scientific field builds on scientific understandings of a person’s unique molecular and genetic profile, helping healthcare providers predict the safest and perhaps most effective course of action and possibly revolutionizing the field of nutritional health
At the Weizmann Institute of Science, two key scientists — Dr. Eran Elinav, an Israeli immunologist and microbiota researcher, and Professor Eran Segal, a computational biologist professor have dedicated their efforts to the microbiome of the gut, an ecosystem that is specific to each and every human being on the planet. Their research served as the basis for the establishment of Israeli startup DayTwo, a company that developed a personalized health platform based on the gut microbiome. They are both on the company’s scientific board.
With the hopes of making advances in the medical field, they are now seeking answers about sustainable, eco-friendly nutrition that would provide personalized benefits for patients.
That Gut Feeling
A recent study led by Dr. Elinav and Professor Segal examined individual-specific responses to diet and its relationship to the gut microbiome –– the ecosystem of all the microorganisms involved in our digestive processes, from the mouth and through the digestive tract. For many of these organisms to thrive, they rely on various nutrients and resources that humans consume from their regular diet. However, a person’s microbiome is also influenced by the composition, amount, and timing of their diet.
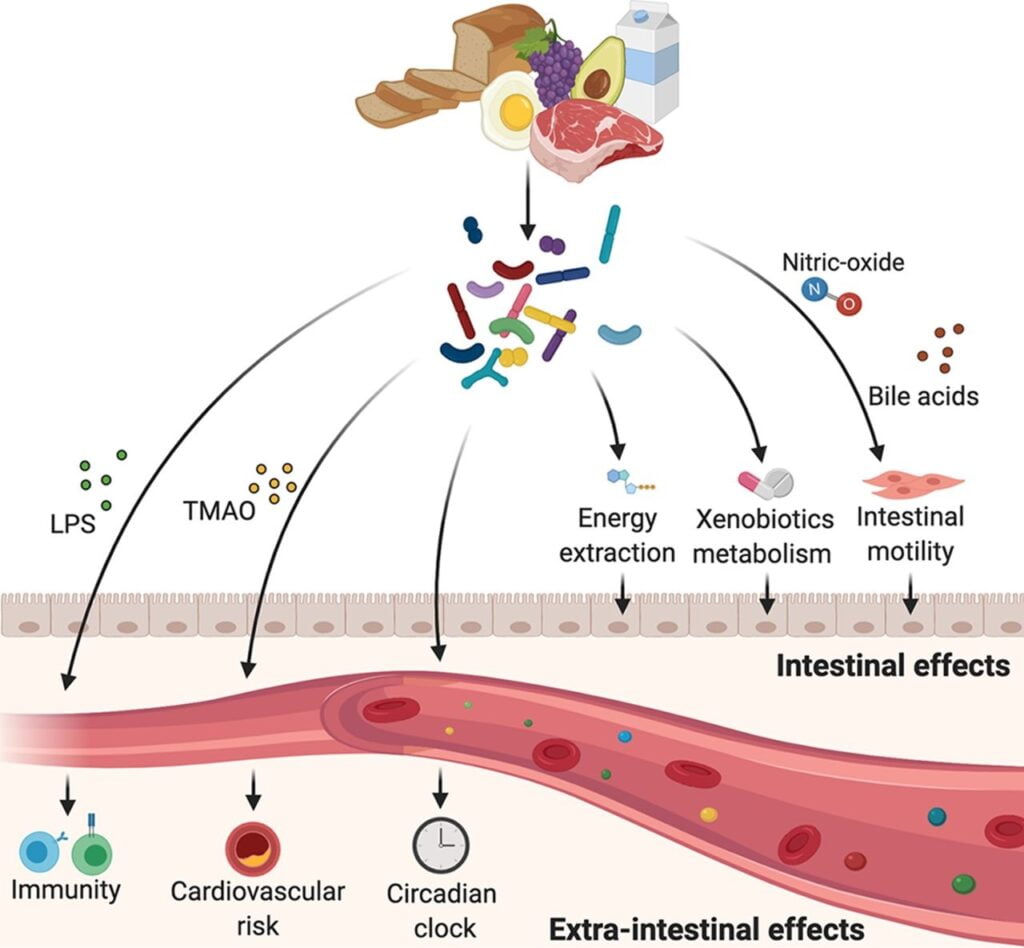
Gut microbiota provides crucial benefits in the form of immune system development, prevention of infections, nutrient acquisition, and even the functionality of the brain and nervous system, and research has shown that it plays a causal role in developing pathologies of obesity, autoimmune diseases, and neurological diseases.
Although a large proponent of one’s gut health originates from genetically inherited traits, the dietary habits and choices we make are essential in establishing the diversity of the gut microbiome. A person’s metabolism, diet, and nutrition can outweigh genetic and environmental factors in determining health outcomes for certain conditions like diabetes, obesity, IBS (irritable bowel syndrome), IBD (inflammatory bowel disease), colorectal cancer, and depression. Therefore, it is just as important to consider the food we consume because our meals more effectively dictate the biology of our gut microbiome.
A Satisfying Scientific Discovery
Because they have significant cardiovascular and diabetic health implications, blood sugar responses of an individual are crucial when determining treatment. With over 400 million people suffering worldwide from diabetes, predicting an individual’s response to food based on their lifestyle and microbiome could expedite successful medical intervention for pre-diabetic patients.
To this end, the Israeli researchers have discovered how a personalized dietary intervention tailored to an individual can substantially improve glycemic responses and parameters.
Sign up for our free weekly newsletter
Subscribe“Our research team had a cohort of about 800 individuals, and we wanted to study their reaction to postprandial –meaning after meals — glycemic responses to several foods. So, we created these standardized meals that consisted of specifically known amounts of calories and carbohydrates,” says Dr. Niv Zmora, MD and PhD from Prof. Elinav’s lab at the Weizmann Institute. “We had each participant log their daily activities, meals, habits and medications for a period of one week. In the end, we analyzed the data and ultimately developed an algorithm to predict postprandial glycemic responses based on individual features.”
The results of their research are dependent on data collected from blood tests, anthropometrics, food diaries, and of course, their gut microbiome.
“Every person gave us a stool sample that we then analyzed and extracted DNA from to understand the composition of their gut microbiota. In the end, we had this algorithm that could predict postprandial glycemic responses more so than the methods used these days like carb counting or calorie counting,” says Dr. Zmora.
After validating the algorithm model, the last step was to devise a real-life menu for the individuals of the study, following up with them for a period of two weeks. One week they were given a ‘bad menu’, one that would induce high postprandial glycemic levels, followed by a ‘good menu’ which would induce mild glycemic responses.
“We saw that people were following our predictions, and we could monitor and maintain normal glycemia, meaning a normal blood glucose level for those following the ‘good menu.’ We also had great data to compare the two,” explained Zmora.
This research is a cornerstone for personalized medicine efforts, laying the groundwork for personalized nutrition. By considering microbiome features, host characteristics, and dietary habits, scientists are able to integrate machine-learning algorithms to further understand how varying individual variability in their gut corresponded to their subsequent sugar levels.
The results of the studies have massive implications for the future prospects of personalized medicine, allowing physicians to predict responses that are tailored to the patient specifically.

“As physicians, we are used to this term ‘personalized medicine,’ and it’s widely practiced. However, it’s still intriguing to see there hasn’t really been an application of personalized nutrition,” says Dr. Zmora. “This is kind of strange because food consists of nutrients, chemicals, and different components that we consume more often than we do medications. It doesn’t quite make sense that we don’t individualize nutrition.”
Not only could a personalized nutritional plan potentially increase patient compliance and prevent weight gain-weight loss, but it can also diminish the risk of evoking detrimental cardiometabolic health outcomes.
SEE ALSO: DayTwo Raises $37M To Help Manage Metabolic Diseases Through Personalized Nutrition
“The major contribution of this study will be to those who struggle to maintain their blood glucose levels in a normal range. This is really good information for pre-diabetics, diabetics, or those suffering from a metabolic condition,” he says. “In the future, we hope to identify the long-term consequences of the gut microbiota considerations for personalized nutrition. Going forward, we hope to expand beyond just metabolism and to explore implications of other pathological medical states like inflammatory states or cancer.”
“Although our study was limited to sugar levels, we think personalized medicine should also include lipid levels, protein metabolism, and other micronutrients. In the end, I think the vision is to be able to devise a comprehensive algorithm for many nutrients and corresponding conditions,” Dr. Zmora concludes.
The findings indicate how health predictions based on one’s microbiome were accurate and clinically beneficial when it came to person-specific responses to certain foods. And how the gut microbiome is at the forefront of scientific discovery, and can pioneer disease-specific treatments and therapies that are more successful and more individualized than current nutrition-counseling efforts.
Related posts

Israeli Medical Technologies That Could Change The World

Harnessing Our Own Bodies For Side Effect-Free Weight Loss

Missing Protein Could Unlock Treatment For Aggressive Lung Cancer




Facebook comments