Israeli-founded firm Sight Diagnostics, the company that uses AI and cutting-edge hardware to deliver rapid, complete blood count results, has signed a new research agreement with Jerusalem’s Shaare Zedek Medical Center to identify the possibility of blood cancer using a combination of images and data collected from Sight’s OLO device and patient clinical information provided by the hospital.
The research focuses on developing the capability to detect and classify white blood cells, and more specifically, to differentiate between different types of white blood cells in the immune system called lymphocytes, a procedure that has greatly challenged the global hematological community, Sight Diagnostics explained in a statement.
Recent publications have indicated that distinguishing between different types of lymphocytes is key to aiding in the early detection of various blood cancers. While reactive lymphocytes appear in the viral infection process, malignant lymphocytes can mean the onset of hematological cancer.

“Differentiating between malignant lymphocytes and reactive lymphocytes is very complex and challenging as both types of lymphocytes may have very similar characteristics. Existing tools and devices involve highly skilled laboratory professionals to carry out complex tasks that often take significant amounts of valuable time, both for the patients and the medical staff,” Barak Bringoltz, Algorithm R&D manager at Sight Diagnostics, tells NoCamels.
“Therefore, the main goal of this new research is to develop algorithms for OLO that can quickly and accurately determine whether the atypical lymphocytes found in the blood samples are reactive or malignant,” he adds.
SEE ALSO: Israel’s Sight Diagnostics To Supply Blood Test Analyzers To Clinics In UAE, Saudi Arabia
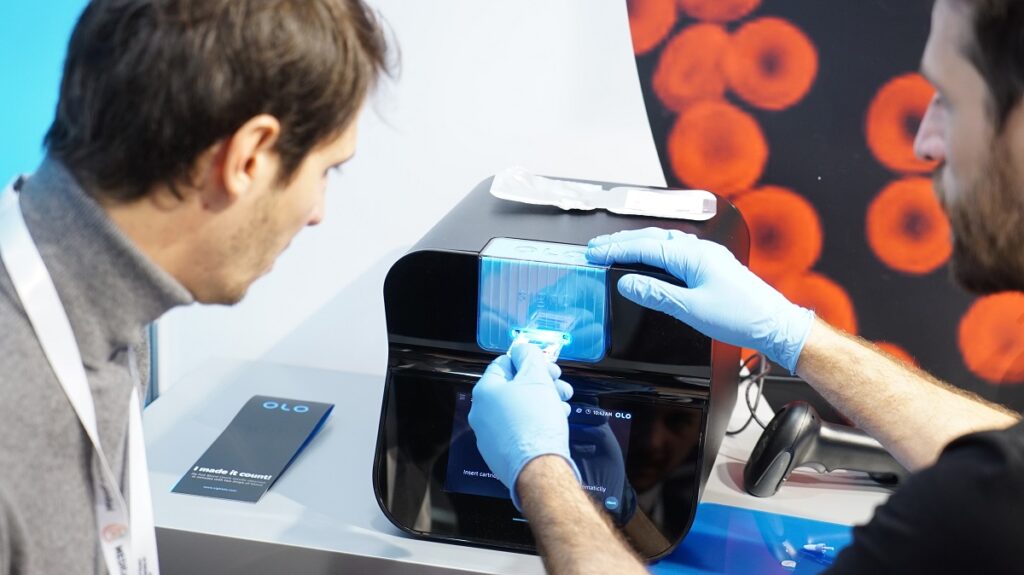
Founded in 2010 by Yossi Pollak and Daniel Levner, Sight Diagnostics uses advanced computer-vision and machine-learning technology in the field of blood diagnostics. The company’s OLO device leverages AI with a revolutionary method to ‘digitize’ blood, allowing patients to receive blood test results within minutes at the point-of-care, and with just a finger-prick. The system takes detailed images of blood and then analyzes them with AI-driven computer vision algorithms. The testing includes CBC (Complete Blood Count) tests which are some of the most basic and informative tests that can be conducted by medical professionals.
The device, about the size of a toaster oven, is compact and uses a cartridge-based system that does not require external reagents or routine calibration and maintenance, making it simple to set-up and operate wherever FBC is needed.
Sight OLO received regulatory approval for use in the UK and is CE Marked according to the IVD European directive at point of care settings. In the United States, Sight OLO is 510(k) cleared for use in moderate complexity laboratories.

R&D manager Bringoltz currently leads Sight’s R&D team as they analyze the images of blood cells collected during the course of the research and employ advanced AI-based to cross-reference the visual information with the clinical data from Shaare Zedek.
“The analysis often starts with detecting intricate patterns in the data, and my role is to direct the team to establish this discovery in careful statistical tests, as well as verifying the phenomenon in additional experiments and on other datasets so that we avoid overfitted modeling and derive the right conclusions,” says Bringoltz. “We are currently in the data collection phase, and the algorithm developers on my team will be tasked with analyzing and modeling the data with machine learning methods once we have collected substantial data from the study.”
When the team has a new algorithm to present, he will help drive the review process, which consists of a forum that includes product specialists, clinical specialists, external experts, and consultants.
Using Sight Diagnostics’ OLO device
The images of blood samples that are part of the research program are captured by OLO instruments at the Sight Diagnostics offices, as well by an OLO instrument placed at Shaare Zedek hospital, Bringoltz tells NoCamels. As part of the research agreement, OLO will collect images of around 150 samples that span across approximately 10 different types of leukemia and lymphoma.
Sign up for our free weekly newsletter
Subscribe“A major piece of information that we obtain from these samples is a clinical diagnosis — what type of cancer it is,” Bringoltz says. By cross-referencing the visual information from the corresponding blood sample with the clinical diagnosis, “we can focus on further investigating features in our imaging data that capture biological changes in the associated malignant blood cells (like the abnormal lymphocytes),” he says.
“The use of OLO in this research is unique in the sense that it can automatically collect over 1,000 different images of the blood sample at different wavelengths of light, different foci, and different positions on the sample,” Bringoltz explains.
Eventually, Sight obtains a digitized version of the blood sample that is represented by approximately 1010 pixels. The company’s algorithm developers and biology experts sift through this data, concentrating on the way lymphocytes appear and look for visual signs that this sample’s lymphocytes features are substantially different from those of other non-malignant samples.
It’s important to note that all the patient data obtained by Sight Diagnostics is de-identified so that team members only know if a blood sample is related to a certain type of malignancy and general patient demographic data, such as age or gender. This is significant because protecting patient’s privacy is a top priority for the research, according to Bringoltz.
Past, present, and future collaborations
Sight Diagnostics first partnered with Shaare Zedek Medical Center two years ago when the hospital launched an innovation center focused on collaborating with digital healthcare companies. The innovation center, called Madait (in Hebrew) and Shaare Zedek Scientific (in English), works with leading tech companies and provides access to samples and data collected at the hospital.
The previous collaboration between Sight Diagnostics and Shaare Zedek focused on exploring the development of a severity index based on the blood image of COVID patients, says Bringoltz. The research examined the morphological changes in the white blood cells of coronavirus patients to provide early detection of COVID-19.
Dr. Renana Ofan, CEO of Shaare Zedek Scientific (Madait) said in a statement that “the long-standing and fruitful collaboration between Shaare Zedek and Sight Diagnostics is an excellent example of our capacity to contribute to product development processes by providing access to data, samples, and our medical staff’s knowledge and expertise, as well as testing the products in real-world clinical settings. Shaare Zedek’s laboratory and clinical teams are working closely with Sight’s R&D team to develop technological solutions that will enable better and faster treatments for patients in Israel and around the world.”
Separately, while Shaare Zedek’s innovation center is not at liberty to disclose details of many of its partnerships with leading tech companies from Israel and around the world, it has partnered with giants like Google, Medtronic, and Japanese multinational information tech firm NEC.

Sight Diagnostics is also in the midst of a number of research collaborations. NoCamels reported last year that Sight deployed its safe blood test technology for coronavirus patients at Israel’s Sheba Medical Center. In December 2020, Sight signed a distribution agreement to supply its OLO analyzers to medical centers in the United Arab Emirates, Saudi Arabia, and other Gulf countries over the next year through Phoenix Capital in Dubai. In March, NoCamels also reported that Sight deployed its OLO analyzer at the John Radcliffe Hospital, a teaching hospital that forms part of the Oxford University Hospitals NHS Foundation Trust.
“We are always looking to improve on both the AI algorithm side and the device hardware side. To date, we have collected close to a petabyte of blood image data, which provides the possibility to implement neural network algorithms that are significantly deeper than those we use today. This can help improve the robustness of OLO’s algorithms to account for varying conditions in the sample’s biology, in the environment, and in the hardware. Beyond that, we are keen to explore the blood images we have accumulated to develop novel algorithms that focus on clinical diagnoses that are linked to abnormal blood morphologies,” explains Bringoltz.
SEE ALSO: How A Portable Blood Test Device Could Spur At-Home Chemo Treatments
“Through the digitization of blood cells, Sight compiles vast amounts of information from each sample, information that can potentially assist in the early detection of diseases and the development of novel tests and diagnostic capabilities,” said Yochay Eshel, Executive VP R&D at Sight Diagnostics, in a company statement. “This research represents another chapter in the productive, well-established collaboration with Shaare Zedek Medical Center and the Madait, and its an important step in our endeavor to continue developing Sight’s blood diagnostic technologies for clinical fields with a visual signature in the blood.”
Related posts

Editors’ & Readers’ Choice: 10 Favorite NoCamels Articles

Forward Facing: What Does The Future Hold For Israeli High-Tech?

Impact Innovation: Israeli Startups That Could Shape Our Future




Facebook comments