Diabetes is a major public health crisis that is approaching epidemic proportions around the globe and growing at an alarming rate. According to the International Diabetes Federation (IDF), over 425 million adults are currently living with diabetes. That number is expected to rise to 629 million by 2045.
The disorder, which occurs when the pancreas does not produce enough insulin (the hormone that regulates blood sugar) or when the body cannot use its produced insulin, has become so deadly, that the International Diabetes Federation said it was responsible for four million deaths in 2017 alone. The World Health Organization estimated diabetes to be the seventh leading cause of death globally in 2016.
Over 350 million people are at risk of developing Type 2 diabetes, when the body becomes to resistant to insulin due to lifestyle facts such as excess weight gain, while more than 1.1 million children are living with Type 1 diabetes, the disorder that occurs when the body’s immune system attacks cells of the pancreas that produce insulin, according to the International Diabetes Federation.
World Diabetes Day, marked annually on November 14 to honor the co-discoverer of insulin Canadian Dr. Frederick Banting, born on that day in 1891, aims to raise awareness of the impact of this disease while promoting its management, care, and prevention. For the past two years (including in 2019,) the theme of World Diabetes Day has been “The Family and Diabetes” promoting the family’s role in awareness and education of the disorder.
To mark the day, NoCamels is taking a closer look at Israel’s role in advancing diabetes research and treatment as well as some of the companies with cutting-edge technology that stand out in the field.
Fighting the epidemic with Israeli innovation
Dr. Irit Yaniv, a general manager at Accelmed Ventures II, a new $100 million venture early-stage health tech fund for medical device and digital health startups and co-founder and chairperson of Type 2 diabetes medical device startup Digma Medical, calls diabetes the “most dangerous” global epidemic. Israel is no exception, she says, citing half a million people living with the disease in the country.

“Additionally, about 300,000 are prediabetic, a condition manifesting as borderline high blood sugar levels and an increased risk to progress to diabetes Type 2 within a few years,” she explains.
Dr. Yaniv says life science entrepreneurs address the challenges presented by the epidemic in a number of ways, including creating companies and projects aimed at reducing the risk of developing the diseases, development of novel drug delivery methods for diabetes drugs, and unique medical devices for managing the disorder.
SEE ALSO: 6 Israeli Companies At The Forefront Of Diabetes Care, Prevention, and Treatment
“There are a few interesting examples such as oral insulin and nasal insulin delivery systems that were developed in Israel,” Yaniv says. A recent success story is Nutrino, [an Israeli] software company for the management of diabetes that was acquired by Medtronic.“
Treatment for Type 2 diabetes has been focused on lifestyle changes and pharmacologic solutions. Both have challenges including patient compliance and adverse effects such as weight gain, hypoglycemia, and other reactions, Yaniv says. More than 50 percent of the patients aren’t able to control the disease, even with combination therapeutics.
“The medical device industry has made many efforts in recent years to address this need,” Yaniv says, “Companies such as GI Dynamics have paved the way for new therapeutic alternatives. Digma Medical has demonstrated initial positive clinical data with its unique duodenal ablation system.”
Yaniv founded Digma Medical in 2013 with Ilan Ben Oren. Backed by leading venture capital firms such as Arkin Holdings and Peregrine Ventures, the company is dedicated to the development of its DiaGone device to treat insulin resistance. DiaGone is an endoscopic, disposable device, that uses innovative laser technology to treat the duodenum, a segment of the small intestine, without an implant. The Duodenal Glycemic Control procedure is a one-time 30 minute GI procedure, in which the gastroenterologist uses DiaGone to treat the duodenum for restoring the natural ability of the body to control glucose levels, said to provide long term remission from Type 2 diabetes and other metabolic syndrome-related diseases.

For Rami Epstein, who assumed the role of CEO at stem cell company Kadimastem in May 2019, “a need exists to find a better solution for the management of diabetes in order to decrease the morbidity, mortality, and costs linked to it and its medical-related care,” he tells NoCamels. This method is more than just controlling insulin levels in the body. It is controlling the dose of insulin administered to the body.
“All Type 1 diabetes and 30 percent of Type 2 diabetes patients depend on the daily administration of insulin in order to control their glucose levels in the blood. This is not ideal since patients have to calculate insulin dosages and take into account meal times and portions, physical activity, and other parameters,” he explains. “Unfortunately, many patients do not manage to stabilize their blood glucose levels properly, thereby risking complications that arise from episodes of hypoglycemia or hyperglycemia,”
Through Kadimastem’s groundbreaking stem-cell therapy technology, developed by the company’s chief scientist Professor Michel Revel at the Weizmann Institute of Science and used as the basis for the company’s founding in 2009, the Ness Ziona-based firm has developed and manufactured an “off-the-shelf” cell product for the treatment of insulin-dependent diabetes based on its proprietary tech platform. The tech platform has been used to treat multiple diseases, including ALS, through the expansion and differentiation of Human Embryonic Stem Cells (hESCs) into clinical-grade functional cells.

The product, called IsletRx, is currently in pre-clinical trials. Its goal is to free patients from continuous monitoring of blood sugar levels and repeated insulin injections. The drug contains an endless source of pancreatic functional islet cells, which produce and secrete insulin and glucagon in response to external glucose levels.
Sign up for our free weekly newsletter
SubscribeMeanwhile, Tel Aviv-based clinical-stage pharmaceutical company Oramed, which is focused on the development of oral drug delivery systems, announced this week that Phase IIb trial evaluating the efficacy and safety of its lead oral insulin candidate, ORMD-0801, has had positive results.
The study was a 90-day, double-blind, randomized, multi-center trial designed to evaluate the safety and efficacy of ORMD-0801 as a treatment for patients with type 2 diabetes, Oramed said in a statement. The primary efficacy endpoint was a reduction in Hemoglobin A1c (A1C, also known as HbA1c, is a key clinical measure of blood glucose control) at Week 12, with no weight gain.
Diabetes and AI
Israeli scientists are taking diabetes management solutions one step further and adding machine learning and AI to the mix. DreaMed Diabetes, a medical tech startup founded in 2014 to develop these types of personalized solutions, announced in September that it had received clearance from the US Food and Drug Administration (FDA) as well as a CE Mark for its DreaMed Advisor Pro, an AI-based insulin dosing decision support software. The software is for patients with Type 1 diabetes using insulin pump therapy with continuous glucose sensors and blood glucose meters (BGMs)

The decision-support platform uses proprietary algorithms to process data from a range of connected devices, including insulin pumps and self-management glucometers. The data is then analyzed to provide an optimized insulin dosing treatment plan to maintain a balanced glucose level.
SEE ALSO: Israeli AI Startup Can Predict Which Diabetes Patients Will develop Kidney Disease
“This clinical and technological advance leverages the power of artificial intelligence to optimize insulin administration in a streamlined and cost-effective manner,” the company said in a statement.
“This year I am more optimistic that a change will happen in how we manage diabetes. We see more technology adopted in all markets, more sharing of data between patients – providers and industry – all for the benefit of offering better care for patients,” the company’s CEO Eran Atlas tells NoCamels in an email. “DreaMed is happy to be part of the leaders in this effort, by offering a unique artificial intelligence technology that can analyze data and recommend in only a few seconds how to optimize the technology. With such technology, the proliferation of expert care can be achieved even in emerging markets.”
Another key player operating in artificial intelligence in the diabetes management market is Sweetch. Founded in 2013, Sweetch offers an AI-based platform that aims to identify those at high risk of developing Type 2 diabetes. The company calls itself the first AI-powered therapeutics solution to help people with the disorder and comes with a mobile app and a wireless Bluetooth-connected scale. The early prevention platform announced last year that it will partner with US-based integrated healthcare system WellSpan Health and provide its app to 15,000 employees, including 200 primary care and specialty physicians, as well as advanced practice clinicians in central Pennsylvania and northern Maryland.
In 2016, the startup raised $3.5 million in a Series A round led by equity crowdfunding platform OurCrowd and Philips.
‘Smart’ sock alleviates symptoms
Diabetes can cause circulation problems and related conditions such as peripheral arterial disease (PAD) which occurs when plaque builds up in the arteries and reduces blood flow to the limbs. For some patients, high levels of blood glucose can damage blood vessels and cause plaque build-up affecting healthy blood flow.
The northern Israel-based startup ElastiMed has developed a wearable medical device that doesn’t treat diabetes patients directly but can help the patient improve his or her circulation.
“It can’t treat the disease directly, but it can treat some of its symptoms,” says Elastimed CEO and founder Omer Zelka.
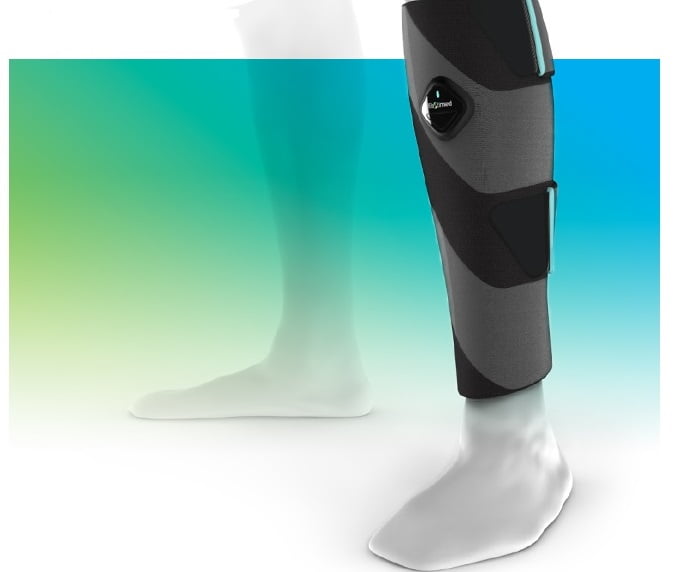
Compression socks improve circulation by squeezing the foot and calf muscles, which straightens out the vein walls to a better working state, says Advanced Tissue, the leading wound care supply provider. Compression therapy is particularly beneficial for diabetes patients because they improve circulation in a non-invasive manner, helping to maintain the right amount of pressure in feet and legs.
ElastiMed’s sock uses battery-operated technology to activate a smart material that compresses and massage the legs to stimulate circulation. The pulses mimic contractions in the calf muscles that in turn increase blood flow.
The sock provides patients with a comfortable, easy-to-wear, highly effective, and cost-effective treatment option to prevent symptoms such as swelling, blood clots, leg ulcers and reduce athletes’ recovery time.
ElastiMed is currently finishing an ongoing clinical study “to demonstrate the safety and the feasibility of this device and its ability to increase venous blood flow.” The study is led by Dr. Vered Shuster Ben-Yosef, R&D Lab Manager of the company and is currently taking place at Hillel Yaffe Medical Center in Hadera. The company currently has a working prototype and aims to get the product on the market in early 2021.
Related posts

Editors’ & Readers’ Choice: 10 Favorite NoCamels Articles

Forward Facing: What Does The Future Hold For Israeli High-Tech?

Impact Innovation: Israeli Startups That Could Shape Our Future




Facebook comments